Reconstituting a peptide for nasal spray research requires precision at every step. One miscalculated concentration, one lapse in sterile technique, and everything built on that preparation becomes unreliable. At Holas, we supply lyophilized research peptides every day, and the questions we get most often are not about the compounds themselves. They are about what happens after the vial arrives.
This peptide reconstitution guide walks through the full process for nasal spray preparation: the equipment you need, the concentration math, the step-by-step mixing technique, and the mistakes that silently compromise results before a single spray is ever administered.
Quick-Reference: Peptide Nasal Spray Concentration Table

Before starting, use this table to identify how much bacteriostatic water to add based on your vial size and target dose per spray. This assumes a standard nasal spray bottle delivering 0.1 mL per actuation.
| Vial Size | Bacteriostatic Water | Concentration | Dose per Spray (0.1 mL) | Total Sprays per Vial |
|---|---|---|---|---|
| 5 mg | 5 mL | 1,000 mcg/mL | 100 mcg | 50 |
| 10 mg | 10 mL | 1,000 mcg/mL | 100 mcg | 100 |
| 10 mg | 5 mL | 2,000 mcg/mL | 200 mcg | 50 |
| 15 mg | 10 mL | 1,500 mcg/mL | 150 mcg | 100 |
| 30 mg | 10 mL | 3,000 mcg/mL | 300 mcg | 100 |
Adjust the volume of bacteriostatic water up or down to hit your protocol’s target dose per actuation. Always confirm your specific bottle’s spray volume before calculating.
Why Nasal Spray Reconstitution Differs from Injection Preparation
Reconstituting a peptide for subcutaneous injection and reconstituting one for nasal spray administration are not the same process. The delivery route changes everything about how you calculate concentration, what diluent you use, and how long your solution remains viable once prepared.
When you prepare a nasal spray, you are creating a multi-use solution. That bottle will be opened, actuated, and recapped repeatedly across days or weeks. Every time that happens, you introduce a new contamination opportunity. A single-use injection vial does not carry that risk. A nasal spray bottle does, and your diluent choice has to account for that from the start.
You also cannot verify absorption the same way you would with an injection. Nasal bioavailability varies depending on formulation, volume per actuation, mucosal contact time, and compound stability in solution. If your concentration is off by even a small margin, your dosing data is off, and you will not know it from the results alone.
Getting reconstitution right for nasal research applications is not optional. It is the foundation on which everything else is built.
Equipment Checklist for Peptide Nasal Spray Preparation
Do not begin without every item accounted for. Leaving the work surface mid-process to retrieve a missing tool is one of the most reliable ways to introduce contamination.
- Lyophilized peptide from a verified supplier with a batch-specific COA confirming purity, identity, and lot number. All Holas peptides ship with independent third-party lab results for every batch.
- Bacteriostatic water containing 0.9% benzyl alcohol, lab-grade, sealed, and within its expiry date
- Sterile syringes, 3 mL or 5 mL, depending on your volume, with 21-gauge needles
- Sterile nasal spray bottle calibrated to deliver a fixed volume per actuation (typically 0.1 mL, confirm before starting)
- Alcohol prep pads, isopropyl at 70% concentration
- Nitrile gloves
- Clean, flat work surface that can be fully disinfected before use
Critical: Confirm the actuation volume before you do any calculations. Bottles marketed as “0.1 mL per spray” are not always exactly that. If your bottle delivers 0.12 mL per actuation and you calculate for 0.1 mL, every dose across your entire study is running 20% higher than your protocol intends. Check the manufacturer specs. Do not assume.
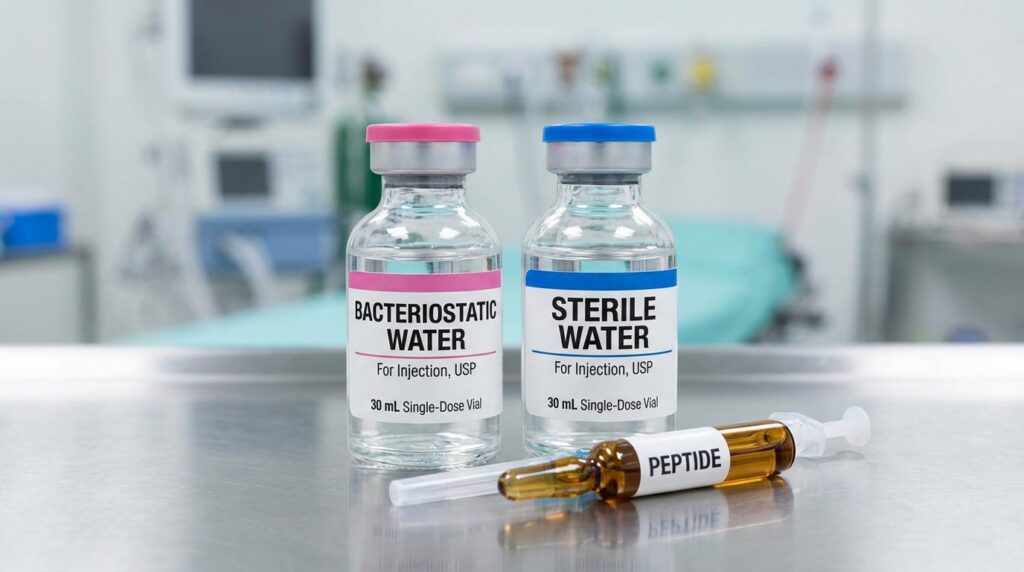
Bacteriostatic Water vs. Sterile Water for Peptide Reconstitution
| Diluent | Preservative | Multi-Use Safe | Reconstituted Shelf Life |
|---|---|---|---|
| Bacteriostatic Water | 0.9% Benzyl Alcohol | Yes | Up to 28 days |
| Sterile Water | None | No | Under 24 hours |
| Saline Solution | None | No | Under 24 hours |
| Tap or Distilled Water | None | Never | Contaminated from start |
For any nasal spray that will be used more than once, bacteriostatic water is the only appropriate diluent.
Sterile water is exactly what the name says: sterile at the moment it is sealed. The moment you open it, that sterility is no longer guaranteed. It has no preservative, which means it has no defense against bacterial growth once introduced into an open system. A nasal spray bottle that sits in a refrigerator between uses, opened and closed across multiple sessions, is precisely the kind of environment where microbial contamination develops without any visible warning.
The 0.9% benzyl alcohol in bacteriostatic water actively inhibits the growth of bacteria throughout the usable life of the solution. That is its entire purpose. Using sterile water for a nasal spray is not a shortcut. It is a protocol error with real consequences for both research validity and safety.
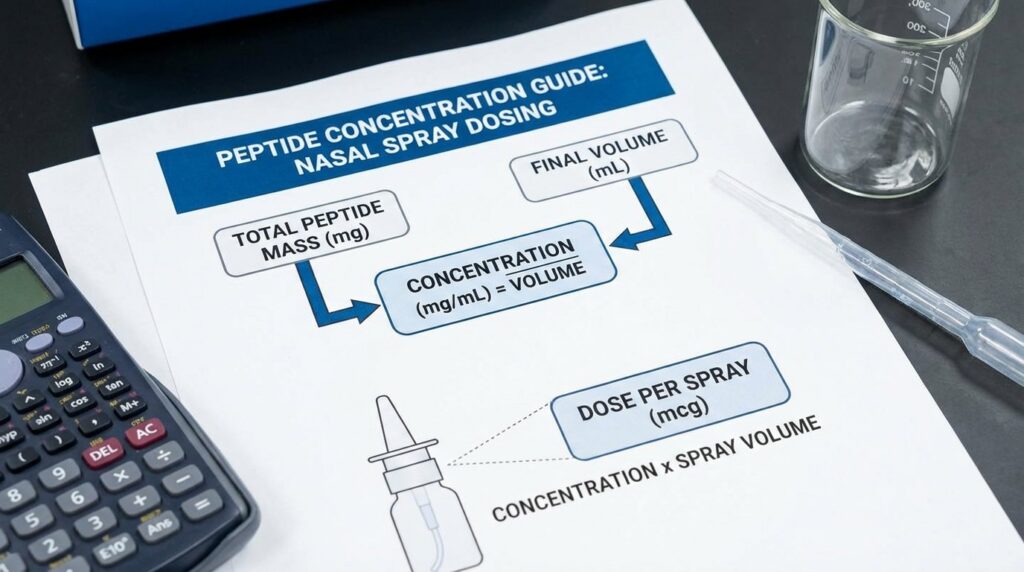
How to Calculate Peptide Concentration for Nasal Spray Dosing
This is where most errors originate. The math is not complicated, but it has to be done before reconstitution begins, written down, and verified twice before you draw a single milliliter into a syringe.
Worked Example
You have a 10 mg vial of lyophilized peptide. Your nasal spray bottle delivers 0.1 mL per actuation. Your research protocol targets 100 mcg per spray.
Step 1: Convert everything to the same unit. 10 mg = 10,000 mcg.
Step 2: Determine the required concentration. If each 0.1 mL spray must contain 100 mcg, then 1 mL of solution must contain 1,000 mcg.
Step 3: Calculate the total volume of bacteriostatic water to add. 10,000 mcg ÷ 1,000 mcg per mL = 10 mL of bacteriostatic water.
Adding 10 mL to your 10 mg vial produces a solution where every 0.1 mL actuation delivers exactly 100 mcg.
Write that number down before you pick up a syringe. Check the math again. A calculation error at this stage cannot be corrected after the fact. Once you add water to the vial, you are committed to that concentration.
Step-by-Step Peptide Reconstitution for Nasal Spray
Step 1: Sanitize Your Work Area and All Materials
Wash your hands thoroughly with soap and water. Put on nitrile gloves. Wipe down the entire work surface with disinfectant and allow it to dry. Lay out every piece of equipment before touching anything else.
Take an alcohol prep pad and wipe the rubber stopper on your peptide vial. Take a fresh pad and wipe the stopper on your bacteriostatic water vial. Let both stoppers air dry for 30 to 60 seconds. The alcohol needs time to fully evaporate and do its work. Wiping a stopper and inserting a needle five seconds later is not sterilization. It is a gesture toward sterilization, and there is a difference.
Step 2: Pressurize the Bacteriostatic Water Vial Before Drawing
Pull the syringe plunger back to the volume of bacteriostatic water you calculated. You are drawing in air, not liquid. Insert the needle through the sanitized rubber stopper of the bacteriostatic water vial without inverting it yet. Push the plunger all the way down, injecting that volume of air into the vial.
What you just did is equalize the pressure inside a sealed vial. Sealed vials have lower internal pressure than the outside environment. Without this step, you are pulling liquid out against a vacuum, which makes accurate volume measurement difficult and introduces more air bubbles into the syringe.
Invert the vial. Draw the bacteriostatic water slowly until you reach your exact target volume. Check for air bubbles. If they are present, flick the barrel of the syringe gently until the bubbles rise to the top, then push them out carefully before withdrawing the needle.
Step 3: Introduce Water Along the Vial Wall (Not Directly onto Powder)
This is the most critical step in the entire process and also the one most commonly done wrong.
Insert the needle through the sanitized stopper of your peptide vial. Do not push the plunger immediately. Angle the needle so the tip is touching the inner glass wall of the vial, not pointing down at the lyophilized powder sitting at the bottom.
Now push the plunger slowly. The bacteriostatic water should trickle down the inside of the glass wall and pool gradually at the base of the vial, wetting the powder from beneath rather than hitting it from above.
The reason this matters: lyophilized peptide is a fragile, porous structure. It looks like a solid cake or compressed powder, but at the molecular level, it is an open lattice held in a specific conformation. A pressurized stream of water blasting directly onto it from above creates shear force. That physical force is enough to disrupt peptide chains before the compound has even begun to dissolve.
Step 4: Mix by Swirling, Never by Shaking
Set the vial on the work surface. Do not shake it.
Shaking a reconstituted peptide solution is one of the fastest ways to degrade what you just carefully prepared. The vigorous back-and-forth motion creates intense shearing forces throughout the liquid and generates foam at the surface. Both are damaging.
Roll the vial slowly between your palms. Swirl it in gentle circular motions. The lyophilized cake will dissolve progressively over the next one to two minutes. Be patient.
When dissolution is complete, the solution should be completely clear. No cloudiness, no visible particles, no color change. If you see cloudiness or particulate matter, do not proceed. Discard the solution, document what you observed, and investigate before starting over.
Step 5: Transfer to the Nasal Spray Bottle
Draw the fully dissolved solution into a clean syringe. Remove the cap from your sterile nasal spray bottle. Transfer the solution directly into the bottle. Make sure the tip of the syringe and the opening of the nasal bottle never contact any surface that has not been sterilized.
Secure the spray cap onto the bottle tightly when transfer is complete.
Step 6: Label the Bottle With Full Documentation
At minimum, your label should include: the name of the compound, the concentration per actuation (in mcg per spray), the total volume in the bottle, the diluent used, and the date of reconstitution.
Store the labeled bottle in the refrigerator at 2 to 8 degrees Celsius. Do not freeze it. For more on proper temperature management, see the guide on storage and handling practices for research peptides.
With bacteriostatic water and proper cold storage, your reconstituted nasal spray is typically stable for up to 28 days from the date of preparation.
Common Peptide Reconstitution Mistakes That Compromise Results
- Shaking the vial instead of swirling. The solution may still look clear after shaking. That appearance does not confirm the compound is structurally intact.
- Using sterile water for a multi-use spray. It holds for the first use. From the second use onward, every actuation introduces unprotected microbial risk.
- Injecting water directly onto the powder. The lyophilized cake is a preserved molecular structure. Hitting it with a pressurized stream does mechanical damage before dissolution even begins.
- Skipping the air injection step before drawing bacteriostatic water. Pulling against a vacuum produces inaccurate volumes and more air bubbles.
- Skipping alcohol prep on vial stoppers. The stopper is the entry point for every needle that touches your compound. It is not sterile by default.
- Store at room temperature between uses. Even a few hours at ambient temperature accelerates peptide degradation. Refrigerate immediately after every use.
- Not confirming the nasal bottle’s actuation volume. This is the assumption that costs you the most when it is wrong.
Reconstitution Guidelines for Specific Research Peptides
The process documented above applies universally across lyophilized peptides. What changes between compounds is the target concentration and sometimes the total volume being prepared. Notes on reconstituting specific peptides available through Holas for nasal spray research:
BPC-157
A pentadecapeptide fragment is commonly studied in tissue repair research. Typical nasal research concentrations range from 100 to 250 mcg per spray. BPC-157 dissolves readily in bacteriostatic water and produces a clear solution without extended mixing time.
Sermorelin
A growth hormone-releasing hormone analog used in GH secretion research. Sermorelin is relatively stable in solution but should be used within the standard 28-day window.
CJC-1295
Another GHRH analog is commonly studied alongside ipamorelin. The non-DAC variant is more commonly used in nasal delivery formats because of its shorter half-life characteristics.
Ipamorelin
A selective growth hormone secretagogue peptide. Ipamorelin reconstitutes cleanly and is stable at standard nasal spray concentrations.
Tesamorelin
A modified GHRH peptide studied in metabolic contexts. Slightly higher vial concentrations (15 mg) mean the reconstitution math differs from 5 mg or 10 mg vials.
Retatrutide
A triple agonist peptide (GLP-1, GIP, glucagon receptor). Given the higher molecular weight and more complex structure, extra care should be taken during the swirl-mixing step.
Semaglutide and Tirzepatide
Both GLP-1 receptor agonists have been studied in metabolic research. These peptides dissolve well in bacteriostatic water but researchers should verify concentration targets carefully, given their potency at low doses.
BPC/TB-500 Blend
Blend vials require the same reconstitution technique. The total peptide weight listed on the vial is the combined weight of both compounds.
How Peptide Quality Affects Reconstitution Outcomes
Not every reconstitution failure traces back to technique. The starting material matters just as much. Before reconstituting any peptide, verify the following from your supplier:
- Batch-specific COA with independent HPLC and mass spectrometry results
- Purity verification at or above the threshold your protocol requires
- Consistent lyophilization with the powder forming a visible cake at the bottom of the vial
Holas publishes lab results for every batch, tested by independent third-party laboratories. Every product ships lyophilized with full batch testing and quality control documentation.
For a deeper look at what separates reliable peptide suppliers from problematic ones, review the key factors when choosing a research peptide supplier. You can also read about warning signs when purchasing peptides online.
Shop Research Peptides at Holas
If you are building a nasal spray research protocol and need laboratory-grade peptides with verified purity and complete documentation, Holas supplies exactly that.
Browse the full catalog or contact us with questions about sourcing the right compound for your work.
Frequently Asked Questions
How long does reconstituted peptide last in a nasal spray bottle?
When reconstituted with bacteriostatic water and stored between 2 and 8 degrees Celsius, most peptide solutions remain stable for up to 28 days. Mark the reconstitution date on the bottle and discard anything past that point, regardless of appearance.
Can you use sterile water instead of bacteriostatic water for a nasal spray?
Not for a multi-use application. Sterile water carries no preservative, which means bacterial growth becomes a risk the moment the bottle is opened. For a nasal spray used across multiple sessions, bacteriostatic water is the only appropriate diluent.
How much bacteriostatic water do I add to a peptide vial?
The amount depends on your target dose per spray and your bottle’s actuation volume. Use this formula: Total BAC water (mL) = Total peptide (mcg) ÷ desired concentration (mcg/mL). For a 10 mg vial targeting 100 mcg per 0.1 mL spray, you need a concentration of 1,000 mcg/mL, so you add 10 mL of bacteriostatic water.
What does the powder in the vial actually contain before water is added?
It is the lyophilized peptide preserved through freeze-drying. Lyophilization removes water from the compound under vacuum while frozen, leaving a porous, dry structure stable for shipping and long-term storage.
What does a correctly reconstituted solution look like?
It should be completely clear with no visible particles, cloudiness, or color. Anything cloudy or with floating material should be discarded immediately.
Is it safe to freeze a reconstituted nasal spray?
No. Freezing causes ice crystal formation that physically disrupts the peptide’s molecular structure. If you need storage beyond 28 days, keep the peptide lyophilized and reconstitute a fresh batch when your protocol resumes.
Can you pre-fill multiple nasal spray bottles from one vial?
Yes, provided your sterile technique is maintained throughout the entire transfer process. Each bottle must be sterile before filling and labeled individually.
What concentration should a peptide nasal spray be?
There is no universal answer. Work backward from your target dose per spray to determine the concentration needed per milliliter, then calculate how much bacteriostatic water to add.
Can you combine two peptides in one nasal spray bottle?
In research settings, combining two reconstituted peptides into a single nasal spray bottle is sometimes done when the compounds are compatible in solution. Reconstitute each peptide separately first, confirm both solutions are clear, then transfer both into the spray bottle.
Why does the solution sometimes look cloudy right after adding water?
Brief cloudiness immediately after introducing the diluent is sometimes normal as the lyophilized structure begins to dissolve. Swirl gently and allow one to two minutes for full dissolution. Persistent cloudiness after complete mixing indicates a problem.
Does the brand or source of bacteriostatic water affect results?
Yes. Bacteriostatic water from unreliable sources may have inconsistent benzyl alcohol concentrations, sterility issues, or undocumented additives.
What happens if you accidentally shake the vial during mixing?
Shaking introduces shearing forces that can denature peptide chains. If shaken aggressively, the conservative decision is to discard the preparation and start with a fresh vial.