Every research peptide you buy arrives as a lyophilized powder sitting at the bottom of a sealed glass vial. Before it is useful for anything, it has to be reconstituted, and the liquid you choose for that job determines whether your compound remains stable for 28 days or begins degrading the moment it leaves the freeze-dried state. At Holas, we supply laboratory-grade research peptides to researchers and academic institutions every day, and the most frequent source of confusion in messages from new customers is not the peptides themselves. It is the water.
This guide covers everything that matters about bacteriostatic water for peptide reconstitution: what it is at a chemical level, why it is the only appropriate diluent for multi-use applications, how to calculate concentrations correctly, the step-by-step technique that preserves peptide integrity, and the specific mistakes that silently compromise results before any research has even begun.
Quick-Reference: Bacteriostatic Water Dilution Chart

Use this table to identify how much bacteriostatic water to add based on your vial size and target concentration. Assumes standard insulin syringe dosing at 0.1 mL per draw.
| Vial Size | Bacteriostatic Water Added | Concentration | Dose per 0.1 mL | Total Doses per Vial |
|---|---|---|---|---|
| 2 mg | 1 mL | 2,000 mcg/mL | 200 mcg | 10 |
| 5 mg | 2 mL | 2,500 mcg/mL | 250 mcg | 20 |
| 5 mg | 5 mL | 1,000 mcg/mL | 100 mcg | 50 |
| 10 mg | 2 mL | 5,000 mcg/mL | 500 mcg | 20 |
| 10 mg | 5 mL | 2,000 mcg/mL | 200 mcg | 50 |
| 10 mg | 10 mL | 1,000 mcg/mL | 100 mcg | 100 |
| 15 mg | 3 mL | 5,000 mcg/mL | 500 mcg | 30 |
Adjust the volume of bacteriostatic water up or down to match your protocol’s target dose. Always confirm the syringe calibration you are working with before calculating.
What Bacteriostatic Water Actually Is
Bacteriostatic water is sterile water for injection that has been combined with 0.9% benzyl alcohol. That small percentage of preservative is what separates it from every other type of water available for laboratory or medical use. The benzyl alcohol does not sterilize the solution in a one-time event. It actively inhibits bacterial growth throughout the usable life of the bottle, which is the entire reason this specific formulation exists.
The term “bacteriostatic” is worth understanding on its own terms. It means the preservative stops bacteria from reproducing. It does not necessarily kill bacteria that are already present. That distinction matters because it tells you what the water is designed for: keeping a previously sterile environment stable across repeated access, not salvaging contaminated material.
For peptide reconstitution, this is the exact property you need. A reconstituted peptide vial will be accessed multiple times over several weeks. Each access point is an opportunity for microbial introduction. The benzyl alcohol suppresses growth at each of those entry points, which is why bacteriostatic water can be used safely for up to 28 days after the first puncture while plain sterile water cannot.
For the chemical reference on benzyl alcohol and its pharmacological profile, the National Library of Medicine’s PubChem database is the authoritative source.
Why Peptides Specifically Require Bacteriostatic Water
Peptides are short chains of amino acids connected by peptide bonds. Those bonds are sensitive to a surprising number of conditions: heat, pH variation, mechanical shear force, oxidation, and microbial contamination. Any one of these can break the chain, and once broken, the peptide loses its structural conformation and its biological activity along with it.
The contamination factor is the one most relevant to reconstitution. When a peptide is reconstituted with a non-preserved diluent and stored in a refrigerator between uses, the liquid becomes a nutrient medium. Bacteria introduced during the initial puncture or any subsequent access point have every condition they need to multiply. Within three to five days, a vial that looks perfectly clear can harbor bacterial populations actively metabolizing and chemically altering the peptide itself.
That is the scenario bacteriostatic water is specifically designed to prevent. The 0.9% benzyl alcohol concentration is not arbitrary. It is the level that reliably suppresses bacterial reproduction without interfering with most peptide structures or producing cytotoxicity at standard research volumes. Injection protocols referenced by the FDA have long recognized this formulation as the standard for multi-dose preparations.
Bacteriostatic Water vs. Other Diluents
| Diluent | Preservative | Multi-Use Safe | Shelf Life Once Opened |
|---|---|---|---|
| Bacteriostatic Water | 0.9% Benzyl Alcohol | Yes | Up to 28 days |
| Sterile Water for Injection | None | No | Under 24 hours |
| Normal Saline (0.9% NaCl) | None | No | Under 24 hours |
| Distilled Water | None | Never | Not sterile to begin with |
| Tap or Filtered Water | None | Never | Contaminated from the start |
For any application where a single vial will be accessed more than once, bacteriostatic water is the only acceptable option. Sterile water is sterile at the moment it is sealed. The second it is punctured, that sterility becomes a countdown rather than a state. Saline carries the same limitation and adds sodium chloride to the equation, which some peptides do not tolerate well.
Distilled, filtered, and tap water are not candidates for consideration at any stage. They are not sterile, they carry minerals and microbes, and using them for peptide reconstitution produces a contaminated solution before the research even begins.
How to Calculate Peptide Concentration
The math is where most reconstitution errors originate. It is not complex, but it must be completed, written down, and verified before any water enters any vial. Once reconstitution is done, the concentration is locked in.
Worked Example
You have a 10 mg vial of lyophilized peptide. Your target dose is 250 mcg per administration, and you are drawing with an insulin syringe at 0.1 mL increments.
Step 1: Convert units so they match. 10 mg = 10,000 mcg.
Step 2: Determine the required concentration. If 0.1 mL must contain 250 mcg, then 1 mL must contain 2,500 mcg.
Step 3: Calculate the total bacteriostatic water to add. 10,000 mcg ÷ 2,500 mcg/mL = 4 mL of bacteriostatic water.
Adding 4 mL to a 10 mg vial produces a solution where every 0.1 mL draw delivers exactly 250 mcg.
Write that calculation down before picking up a syringe. Verify it twice. A mistake at this stage cannot be undone after the water has been added.
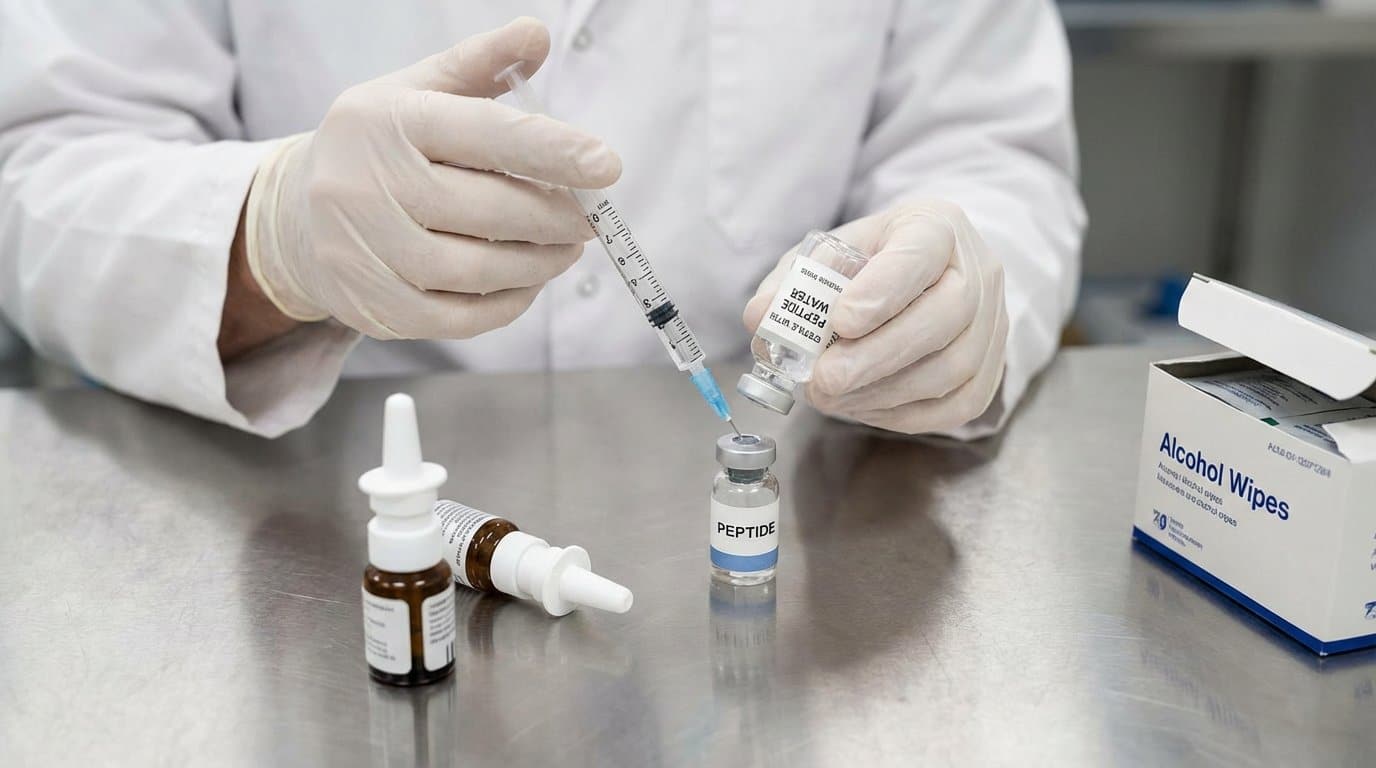
Step-by-Step Reconstitution Protocol
Step 1: Prepare the Work Surface and Materials
Wash your hands thoroughly with soap and water. Put on nitrile gloves. Disinfect the work surface and allow it to dry. Lay out every item needed for the procedure before starting: peptide vial, bacteriostatic water, insulin syringe, and alcohol prep pads. Leaving the workspace mid-procedure to retrieve a missing item is one of the most common contamination vectors.
Step 2: Sanitize the Vial Stoppers
Wipe the rubber stopper on the peptide vial with a fresh alcohol prep pad. Use a separate pad for the bacteriostatic water vial. Allow both stoppers to air dry for 30 to 60 seconds. This dry time is not optional. Alcohol needs to evaporate completely to complete the sterilization process. Inserting a needle into a wet stopper drives residual contaminants directly into the vial.
Step 3: Pressurize the Bacteriostatic Water Vial
Pull the syringe plunger back to the volume of bacteriostatic water you calculated, drawing air into the syringe. Insert the needle into the sanitized stopper of the bacteriostatic water vial without inverting it. Push the plunger down to inject the air into the vial.
This step equalizes the pressure inside the sealed vial. Sealed vials hold slightly negative pressure relative to the outside environment. Without equalizing, drawing water requires pulling against a vacuum, which produces inaccurate volumes and traps air bubbles in the syringe.
Step 4: Draw the Bacteriostatic Water
Invert the vial. Draw the bacteriostatic water slowly until the syringe reaches the exact target volume. Check for air bubbles in the barrel. If bubbles are present, tap the side of the syringe gently until they rise to the top, then push them out before withdrawing the needle.
Step 5: Transfer Water Along the Vial Wall
This is the most critical step in the process and the one most often done incorrectly.
Insert the needle into the sanitized stopper of the peptide vial. Before pushing the plunger, angle the needle so the tip rests against the inner glass wall of the vial rather than pointing down at the lyophilized powder. Push the plunger slowly. The bacteriostatic water should run down the inside of the glass wall and pool at the base of the vial, wetting the powder from underneath rather than striking it from above.
The reason this technique matters: lyophilized peptide is not simply dry powder. It is a porous molecular lattice preserved in a specific conformation through the freeze-drying process. A pressurized stream of water hitting that structure directly creates shear force sufficient to damage peptide chains before they begin to dissolve. Introducing water along the wall allows gentle, gradual dissolution that preserves molecular integrity.
Step 6: Mix by Swirling, Never by Shaking
Place the vial on the work surface. Do not shake it under any circumstances.
Shaking generates shear forces and produces foam at the surface of the solution. Both degrade peptide structure. The damage may not be visible, the solution can look completely clear after aggressive shaking, but the biological activity has been compromised.
Roll the vial gently between your palms or swirl it in slow circles. Dissolution typically takes one to two minutes for standard peptide concentrations. When fully dissolved, the solution should be completely clear with no visible particles, cloudiness, or color. Cloudy solutions or those with particulate matter should be discarded rather than used.
Step 7: Label and Store
Label the vial immediately with the compound name, concentration, date of reconstitution, and diluent used. Store between 2°C and 8°C (36°F to 46°F) in a refrigerator. Do not freeze. For more general guidance on aseptic technique, the CDC injection safety page is a useful reference.
Under proper refrigerated storage, reconstituted peptides typically remain stable for up to 28 days. Some specific compounds have shorter or longer windows, consult your compound documentation.
Common Reconstitution Mistakes
Shaking instead of swirling. The solution can appear clear after shaking. That appearance does not confirm structural integrity. Denatured peptides look identical to intact ones in solution.
Using sterile water for a multi-use vial. It holds for the first 24 hours. After that, microbial risk escalates every time the vial is accessed.
Blasting water directly onto the lyophilized cake. Mechanical damage occurs before dissolution. This is preventable with a 10-second technique adjustment.
Skipping the air injection step. Pulling against a vacuum produces inaccurate volumes and introduces bubbles that affect subsequent measurements.
Not allowing alcohol prep to dry. Wet stoppers transfer residual contaminants directly into the vial.
Storing at room temperature between uses. Even a few hours at ambient temperature accelerates degradation. Refrigerate immediately after every access.
Using expired bacteriostatic water. Once opened, the 28-day window applies regardless of how the water looks.
Reusing syringes across vials. Fresh needle for every access point, without exception.
How to Identify Quality Bacteriostatic Water
Not all bacteriostatic water meets research-grade standards. Before purchasing, verify:
- Manufactured in an FDA-registered facility
- Clearly labeled with lot numbers and expiration dates
- Tamper-evident packaging with an intact seal
- Exactly 0.9% benzyl alcohol concentration specified on the label
- Sourced from a supplier that specializes in research-grade materials rather than general consumer supplies
Pricing alone is not a reliable quality signal. Some reputable suppliers offer competitive prices through efficient distribution. Some lower-quality sources charge premium prices assuming buyers associate cost with quality. Verify credentials and read supplier reviews before committing to a vendor.
Safety Considerations
Bacteriostatic water is formulated for research and multi-dose medical applications in adult populations. It is specifically not appropriate for neonatal use, where benzyl alcohol exposure can produce serious adverse effects. For reference material on medication components and interactions, MedlinePlus provides reliable background.
All peptide research carries responsibilities beyond technique. Use peptides only for legitimate research purposes, maintain detailed protocol records, and follow any applicable institutional review requirements. Treat every vial as what it is: a biologically active compound that deserves careful handling and appropriate respect for its intended use.
Shop Bacteriostatic Water and Research Peptides at Holas
If you need laboratory-grade bacteriostatic water alongside verified research peptides with complete documentation, Holas supplies exactly that. We stock Pfizer Bacteriostatic Water, USP, a sterile, non-pyrogenic 30 mL multi-dose vial with 0.9% benzyl alcohol, ready for research use and shipped with the same care as every peptide in our catalog. Every batch of research peptides we supply is backed by independent third-party lab results for purity and consistency verification.
Browse the full catalog, explore our research peptide bundles, or contact us with questions about sourcing the right materials for your research.
Frequently Asked Questions
Can I use sterile water instead of bacteriostatic water for peptide reconstitution? How long does bacteriostatic water last once opened?
Only for single-use applications consumed within 24 hours. For any multi-dose vial accessed over days or weeks, sterile water cannot provide the preservation needed and bacteriostatic water is the only appropriate choice.
How much bacteriostatic water do I add to a peptide vial?
The amount depends on your target concentration. Use this formula: Total BAC water (mL) = Total peptide (mcg) ÷ desired concentration (mcg/mL). For a 5 mg vial targeting 250 mcg per 0.1 mL, you need 2,500 mcg/mL, so you add 2 mL of bacteriostatic water.
Why does my peptide look cloudy right after adding water?
Brief cloudiness during initial dissolution is sometimes normal as the lyophilized structure breaks apart. Swirl gently and allow one to two minutes for complete mixing. If cloudiness persists after full dissolution, or if visible particles remain, discard the solution.
Can I freeze reconstituted peptides for longer storage?
Most reconstituted peptides should not be frozen. Ice crystal formation physically disrupts peptide molecular structure. If storage beyond 28 days is needed, keep the peptide lyophilized and reconstitute a fresh batch when needed.
Is bacteriostatic water the same as saline?
No. Saline contains sodium chloride and no preservative. Bacteriostatic water contains 0.9% benzyl alcohol and no salt. They are not interchangeable for peptide reconstitution.
What happens if I accidentally shake the vial during mixing?
Shaking introduces shearing forces that can denature peptide chains. The solution may still look clear, but biological activity can be reduced or eliminated. If aggressive shaking has occurred, the conservative approach is to document it and consider the vial compromised for research purposes.
Can I use bacteriostatic water on all research peptides?
The vast majority of research peptides reconstitute well with bacteriostatic water. A small number of peptides are sensitive to benzyl alcohol and require sterile water for immediate single-use applications. Always verify compatibility against your specific compound’s documentation.
Do I need a prescription to buy bacteriostatic water?
Regulations vary by jurisdiction. In many regions, bacteriostatic water is available through research supply channels without a prescription. Check local rules before ordering.
How do I know if my reconstituted peptide has gone bad?
Signs of degradation include persistent cloudiness, discoloration, visible particles, unusual odors, or reduced activity compared to fresh batches. When in doubt, discard and reconstitute a new vial.
Does the source of bacteriostatic water affect results?
Yes. Bacteriostatic water from unreliable sources may have inconsistent benzyl alcohol concentrations, sterility failures, or undocumented additives. Source only from suppliers with verifiable quality control documentation.
Can I reuse a syringe on the same vial?
No. Fresh sterile syringe for every access point, without exception. Reusing syringes introduces contamination risk that the bacteriostatic water’s preservative cannot fully mitigate across extended use.